
Hummingbird Scientific has supported and collaborated in a study led by Stanford University, conducted at the Lawrence Berkeley National Lab’s Advanced Light Source that peers into the electrochemical reactions that fuel common rechargeable batteries of today, revealing new understanding at the nanoscale level. By developing an operando X-ray microscope liquid-electrochemistry platform, researchers at Stanford, MIT, LBL and Hummingbird Scientific tracked the evolution of compositionally nonuniform solid-solution domains during charging and discharging of LiXFePO4 battery particles. For researchers hoping to improve today’s battery performance, counteracting these detrimental forces to the delithiation (charging) and lithiation (discharging) processes could lead to new battery designs and power management for future battery generations. These findings were published in the latest issue of Science.

Using Hummingbird Scientific liquid cell technology, Dr. William Chueh and his team built a transparent battery consisting of two silicon nitride transparent windows with a very small gap between the microfabricated chips through which electrolyte flowed to deliver the lithium ions to the nanoparticles, creating the nano-battery.
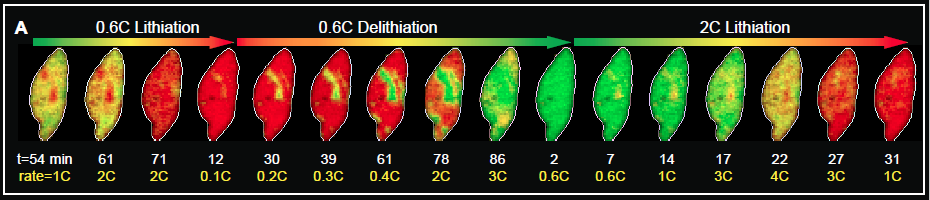
This data showed that spatial heterogeneities in reaction rates account for the domains with the charging process significantly less uniform than the discharging process. These results highlight the crucial role of surface reaction rate for lithiation, observing these inconsistencies of ion insertion have implications for electrode engineering and battery management for future generation battery technology. Jongwoo Lim, a co-lead author of the paper and post-doctoral researcher in the Stanford Institute for Materials & Energy Sciences at SLAC states in Stanford press release that “It gives us fundamental insights into how batteries work. Previously, most studies investigates the average behavior of the whole battery. Now, we can see and understand how individual battery particles charge and discharge.” This could result in batteries that charge faster and more fully than current models.
The instrumentation developed in this project allows researchers to operate microscale batteries in such a way as to be amenable to characterization with ultrabright synchrotron light sources, and builds upon ten years’ worth of research and development of similar instruments for transmission electron microscopes. Funding for these developments have come from the Small Business Innovation and Research programs, with support from the Department of Energy, the National Institutes of Health and the Department of Defense, and the Washington State Department of Commerce for both these developments and for prior, related work. Microfabrication capabilities at the University of Washington’s Nanofabrication Facility were utilized in the research. The resulting instrumentation allows multimodal characterization of battery electrodes via both synchrotron radiation and electron microscopy studies, and is commercially available.
Learn more about the X-Ray Liquid-Electrochemical Cell used in the experiment.
Related product In-situ TEM Liquid-Electrochemistry holder
Link to LBL feature article on this study
Link to Stanford press release
Reference: “Origin and hysteresis of lithium compositional spatiodynamics within battery primary particles”, J. Lim,Y. Li, D. H. Alsem, H. So, S. C. Lee, P. Bai, D.A. Cogswell, X. Liu, N. Jin, Y. Yu, N. J. Salmon, D. A. Shapiro, M. Z. Bazant, T.Tyliszczak, W. C. Chueh, Science 05 Aug 2016 Abstract
View All News

